Diabetes Technology Moves Closer To Making Life Easier For Patients

Pricking your fingers may someday be a thing of the past for diabetics as new technologies aim to make blood sugar regulation more convenient.
Alden Chadwick/Getty Images
hide caption
toggle caption
Alden Chadwick/Getty Images
For people with diabetes, keeping blood sugar levels in a normal range – not too high or too low – is a lifelong challenge. New technologies to ease the burden are emerging rapidly, but insurance reimbursement challenges, supply shortages, and shifting competition make it tough for patients to access them quickly.
One new product is a fast-acting insulin from Novo Nordisk. It is designed to help to minimize the high blood sugar spikes that often occur when people with diabetes eat a meal containing carbohydrates.
This new formulation, branded “Fiasp,” adds niacinamide (vitamin B3), which roughly doubles the speed of initial insulin absorption compared to current fast-acting insulins taken at mealtime. This new insulin hits the bloodstream in under three minutes.
Another advance is Abbott’s new monitoring device called the FreeStyle Libre Flash. It’s new in the U.S. but has been available in Europe since 2014. It’s a round patch with a catheter that is inserted on the arm for up to 10 days and a durable scanning device that the user waves over the patch to read their blood sugar level.

The FreeStyle Libre Flash lets users monitor blood glucose levels without having to frequently prick their fingers for blood testing.
Abbott
hide caption
toggle caption
Abbott
The Libre works a bit differently than the two currently available continuous glucose monitors (CGMs) made by Dexcom and Medtronic. The Libre doesn’t require users to prick their fingers for blood tests to calibrate it, whereas users of the other monitors must perform twice-daily fingerstick calibrations.
Also, the Libre is approved for longer wear – 10 days (14 in Europe) versus seven days for the two current CGMs. And, it is likely to be considerably less expensive, although Abbott isn’t providing cost information for the U.S. just yet. In Europe, the Libre system costs about four Euros a day (about $4.70).
But, unlike the current devices, the Libre doesn’t issue alarms to users when their blood glucose levels get too high or too low. And the U.S. version also doesn’t allow for the “share” capability, by which loved ones can follow Dexcom glucose monitor users’ blood sugar levels remotely via a smartphone app.
The Libre has been extremely popular in Europe among people with type 1 diabetes. There, fewer people use traditional CGMs compared to the U.S., in large part because they are not frequently covered by European insurance.
Type 1 diabetes requires regular insulin doses to allow cells to use glucose, because the pancreas does not make any of its own. With type 2 diabetes, the insulin being made doesn’t adequately meet the body’s needs.
About a quarter of people with type 2 diabetes take insulin, and of those, a smaller number take fast-acting insulin before meals. Those doses can lead to low or high blood glucose levels if not matched perfectly by timing and amount to the meal’s carbohydrates. There’s lots of room for user error.
University of California, Los Angeles endocrinologist Dr. David T. Ahn, who specializes in diabetes technology, believes that in the U.S., the Libre will be more useful for people with type 2 diabetes. Most people with type 2 do not use CGMs and may also not perform frequent fingerstick checks.
“I think it’s something that really empowers people, and that’s what’s really exciting, Ahn says. “[Y]ou literally see firsthand what exercise, diet, rest, and stress do to your blood sugars.”
Of course, he adds, “There’s benefit really for everybody, but the most important question is where is the cost justified. I would say that at least right now, it probably is only worth the cost for someone on insulin, especially on fast-acting insulin.”
Jared Watkin president of Abbott Diabetes Care Division, tells Shots that the Libre was designed for people with either type of diabetes who require frequent glucose testing, and the lack of alarms was intentional. Research shows “alarm fatigue” is one of two main reasons many patients mention for not wanting to use CGM systems, he says. The other reason is cost.
He points out that research on the Libre has also shown that people using the device achieve better glucose control and experience fewer low blood sugar episodes overnight compared to fingersticks alone even without the alarms, presumably because they’re making more insulin dose adjustments.
Aaron Kowalski is the chief mission officer for JDRF, formerly the Juvenile Diabetes Research Foundation, which funds much of the research into diabetes technologies. He says it will be interesting to see how U.S. patients with type 1 diabetes who haven’t adopted CGM take to the Libre. “If you’re coming at it from fingersticking, it makes massive sense … For some people with type 1, I think it will be a really good option.”
Closing the Loop: Progress And Pitfalls
Both continuous glucose sensing and fast-acting insulin are critical components to the development of so-called “closed-loop” or artificial pancreas systems, which aim to automate insulin delivery to the point that patients themselves don’t need to make complicated and error-prone calculations about how many carbs are in their meals or how much to cut back their insulin doses for exercise.
In September 2016, The U.S. Food and Drug Administration approved Medtronic’s 670G, the first device that partially accomplishes the closed-loop goal via an algorithm that allows the system’s CGM to instruct its insulin pump to cut off delivery if the user’s blood sugar drops, or increase it if the levels go too high.
Several other companies are working on similar technology. One of those, a start-up called Bigfoot Biomedical is working with Abbott to use a next-generation version of the Libre’s sensor. Except for Medtronic, the other major closed-loop competitors – Insulet, Tandem, and Beta-Bionics – are all collaborating with Dexcom.
At the same time, a group of do-it-yourself hackers has figured out how to create their own closed-loop systems using older equipment and instructions that are freely available. Since that endeavor isn’t regulated by the FDA, people who have done it – believed to number in the thousands at this point – proceed at their own risk. No major problems have been reported.
Bumps in the Road, But Optimism Overall
As might be expected, not everything in this field has gone smoothly. Due to both high demand and the fact that one of Medtronic’s manufacturing plants located in Puerto Rico was damaged by Hurricane Maria, it has been unable to ship part of the 670G to new users, and may not be able to meet demand until 2018.
In addition, the insurance company Anthem has said it won’t cover the 670G because it has concluded “there is not yet enough data on the longer-term safety and efficacy” for the system.
Meanwhile, although Medicare agreed in January 2017 to cover the Dexcom continuous glucose monitor for beneficiaries who use insulin, the agency recently determined that the device would not be covered if beneficiaries use the accompanying smartphone app that reads the glucose levels via bluetooth, because it doesn’t meet the definition for “durable medical equipment.”
The decision means that seniors have to carry around a separate receiver device, and don’t have access to the share function. Dexcom is negotiating with federal regulators to work out a solution. In a recent blog post, Ahn wrote “While CGM approval by Dexcom is a huge win overall (it really is), restricting smartphone integration is absolutely ridiculous.”
In another blow to the diabetes technology world, major pump manufacturer Animus recently announced that it was pulling out of the market and is shifting its approximately 90,000 current customers to Medtronic. Not surprisingly, some of Medtronic’s competitors are offering deals to lure them to their own products.
Despite the roadblocks, Kowalski says, “I have tremendous optimism about the future for people with type 1 diabetes. These tools are really starting to … improving blood sugar and making life easier. And that’s a great thing. The more options the better.”
Miriam E. Tucker is a freelance journalist specializing in medicine and health. You can follow her on Twitter: @MiriamETucker
Draft Of Health Care Bill Addresses Trump Concerns About 'Bailouts' For Insurers

Sen. Patty Murray, D-Wash., and Sen. Lamar Alexander, R-Tenn., leaders of the Senate Health, Education, Labor, and Pensions Committee, meet before the start of a hearing on Capitol Hill.
J. Scott Applewhite/AP
hide caption
toggle caption
J. Scott Applewhite/AP
Updated at 4:06 p.m. ET
A proposal in the Senate to help stabilize Affordable Care Act marketplaces would ensure that subsidies paid to insurance companies benefit consumers rather than padding the companies’ profits.
A draft of the bill, obtained by NPR, requires health plans to offer the subsidies as one-time or monthly rebates to consumers or they will be repaid to the federal government. The subsidies, known as cost-sharing reduction payments, are designed to reimburse insurance companies for discounts they are required to offer their customers on copayments and deductibles. President Trump has criticized the payments as a “bailout” and said last week he would cut them off.
The bipartisan bill, proposed by Sens. Lamar Alexander, R-Tenn., and Patty Murray, D-Wash., restores the payments for the rest of this year and the next two years. But the lawmakers are seeking to reassure the president that the payments will benefit consumers and not insurance companies.
Trump told reporters at the White House Wednesday morning that he was looking forward to seeing the bill. White House press secretary Sarah Huckabee Sanders told reporters on Wednesday afternoon that Trump does not support the deal in its current form.
“If something can happen, that’s fine. But I won’t do anything to enrich the insurance companiesbecause right now the insurance companies are being enriched. They’ve been enriched by Obamacare like nothing anybody has ever seen before. I am not going to do anything to enrich the insurance companies,” Trump said.
He had tweeted such concerns earlier on Wednesday.
I am supportive of Lamar as a person & also of the process, but I can never support bailing out ins co’s who have made a fortune w/ O’Care.
— Donald J. Trump (@realDonaldTrump) October 18, 2017
Alexander tells NPR’s Susan Davis that he has spoken to the president three times in the past 12 days, including Wednesday morning before the president’s tweet. He also reiterated that the effort to repeal and replace the Affordable Care Act would not end because of this bill.
“We would like to replace Obamacare, that’s almost unanimous in the Republican caucus. But I think it will become increasingly unanimous in the Republican caucus that we understand that between today and the time we can replace Obamacare is two to three years,” Alexander told NPR. “And that if we do nothing we’ll create chaos, and chaos will lead to a birthday present for Bernie Sanders, which is a single-payer solution — which none of us want.”
Murray and Alexander are expected to officially release the bill with a list of bipartisan co-sponsors on Thursday. Republicans are working a number of influential senators from the moderate and conservative wings to sign on so the bill can pass the Senate.
Republican Senate leaders have indicated they will not waste limited floor time on another failed health care bill, which means the bill must be ensured 60 votes to pass.
Senate Minority Leader Chuck Schumer, D-N.Y., criticized the president at length for tempering his initial support for the deal. “This president keeps zigging and zagging so it’s impossible to govern,” Schumer said on the Senate floor Wednesday morning. “He’s totally inconsistent. For it one day, against it the next day. You can’t govern — Mr. President, you cannot govern a country, you cannot keep America great, if you don’t know what’s in the bills and you don’t have a consistent policy about them.”
Schumer said he’d had private conversations with the president in recent weeks in which they both agreed to encourage their respective senators to reach a deal.
The bill also allows states to seek waivers to create variations on the Affordable Care Act in their own states. That’s been a priority of Republican lawmakers, who argue that state-level legislators and governors better understand the needs of their citizens.
“This will give states meaningful flexibility,” Alexander said Tuesday in discussing the forthcoming bill.
The ACA allows states to apply for waivers to set up systems such as reinsurance programs that protect insurance companies from large and unexpected losses, or high-risk pools to provide coverage to the sickest patients.
But many of the state applications have been delayed or denied because of all the requirements laid out in the law.
For example, the ACA requires state legislatures to pass a law approving the waiver provisions. The new proposal would simply allow a state governor to sign off on the plan.
The bill also restores some of the federal budget for advertising and outreach for open enrollment in ACA health plans, which starts on Nov. 1. The Trump administration slashed that budget by 90 percent.
Alexander says he will seek Senate co-sponsors for the bill and then bring it to Majority Leader Mitch McConnell later this week for consideration.
NPR’s Susan Davis contributed to this report.
Full text of the bill and a summary are below.
Drug Companies Make Eyedrops Too Big, And You Pay For The Waste

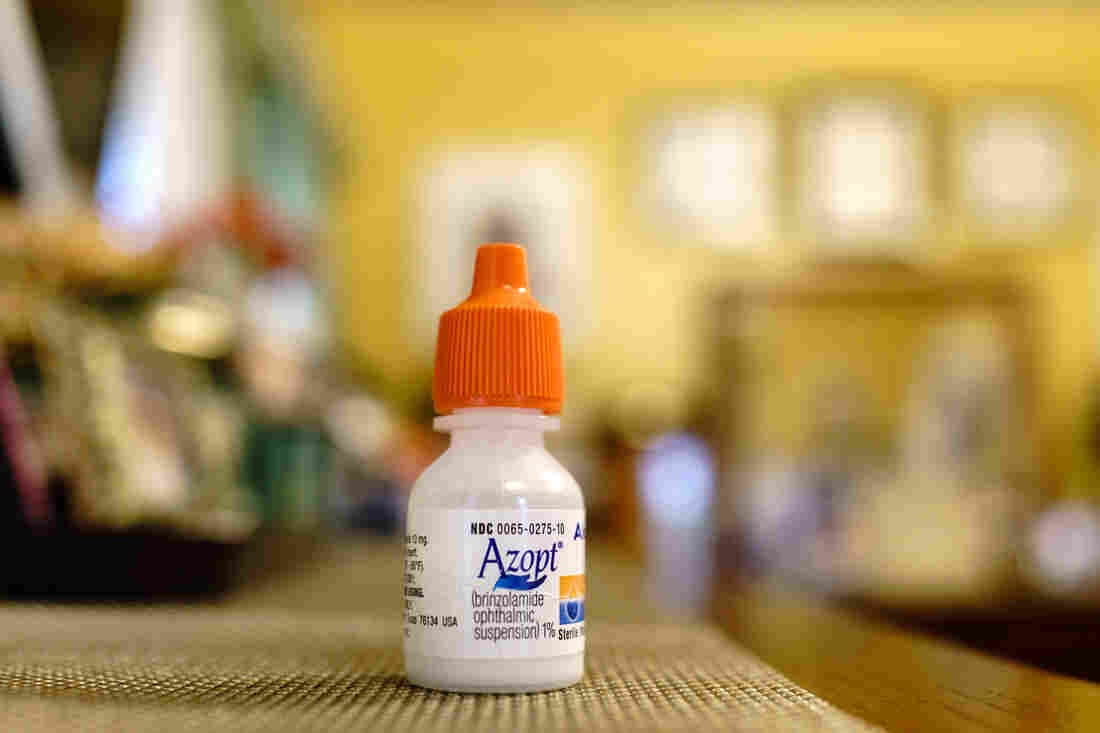
Gregory Matthews has glaucoma and uses prescription eyedrops. The dropper’s opening creates a bigger drop than he needs, causing him to run out of his medication before the prescription is ready to refill.
Matt Roth for ProPublica
hide caption
toggle caption
Matt Roth for ProPublica
If you’ve ever put in eyedrops, some of them have almost certainly spilled onto your eyelid or cheek.
The good news is the mess doesn’t necessarily mean you missed. The bad news is that medicine you wiped off your face is wasted by design — and it’s well-known to the drug companies that make the drops.
Eyedrops overflow our eyes because drug companies make the typical drop – from glaucoma drugs that cost hundreds of dollars to a cheap over-the-counter bottles – larger than a human eye can hold. Some are so large, that if they were pills, every time you swallowed one, you’d toss another in the garbage.
The waste frustrates glaucoma specialists like Dr. Alan Robin, whose patients struggle to make pricey bottles of drops last. He has urged drug companies to move to smaller drops – to no avail.
“They had no interest in people, their pocketbooks or what the cost of drugs meant,” said Robin, a Baltimore ophthalmologist and adjunct professor at the University of Michigan Medical School.
ProPublica has been documenting the many ways money is wasted in health care. We’ve shown how hospitals throw out brand new supplies, nursing homes flush tons of unexpired medication and drug companies concoct costly combinations of cheap drugs. Recently we described how arbitrary expiration dates for drugs cause us to toss safe and potent medicine.
Often, large swaths of the medical and pharmaceutical communities know about this waste—even about solutions to it—but do nothing. Those who end up paying the bill, in one way or another, are consumers.
Crucial eye medications to treat conditions like glaucoma may cost hundreds of dollars for a small bottle that only lasts a month, making the waste of even a drop a problem for low-income patients. Last year, drug companies brought in about $3.4 billion in the U.S. alone on drops for dry eyes and glaucoma drops, according to the research firm Market Scope.
Gregory Matthews said there have been times when he’s run out of his $295 bottle of Azopt, a glaucoma medication, with a few days remaining before his refill and he’s blamed himself. “You feel like you’re doing something that’s going to cause your blindness and it’s because of you,” said Matthews, 63, a teacher from Baltimore.
Smaller Drops Are Feasible
Pharmaceutical companies have done research showing that it’s possible to waste less – and save consumers money. Some of that research has been around for decades.
Robin, for example, consulted in the early 1990s with Alcon Laboratories, one of the world’s largest eye care companies, when its researchers developed a so-called microdrop. Patients, he said, were able to safely and effectively deliver the tiny drops, with nothing wasted. But instead of being a breakthrough, the innovation, he said, became a case study in how business interests trump patient needs.
In the early 1990s, Bill York recalled his bosses at Alcon coming to him with a pressing request. Patients were complaining that some of the company’s drops caused stinging and burning in their eyes. Could he find a fix?
York, head of the research lab at the company’s Fort Worth, Texas, headquarters, knew one way to ease the irritation: Make the drops smaller. The size of eyedrops isn’t regulated, he said recently. Some are over 50 microliters, more than twice what the eye can hold.
When drops are too big, the overflow runs down the face or drains into the body through the ducts in the corner of the eye, he said. This explains why you sometimes get the sensation of “tasting” eyedrops – they’ve entered your sinuses.
“If it spills out, it’s just wasted,” said York, who has a doctorate in pharmaceutical chemistry and is now retired. “It’s not doing any good.”
So his team created a 16-microliter drop — a microdrop — that was about a half to a third of the size of most drops on the market today, he said. The team used a standard bottle with a latex dropper tip that wouldn’t cause injury if it touched the eye. Then they recruited 29 glaucoma patients to test the tiny drops. Glaucoma, a leading cause of blindness in the United States, is characterized by increased pressure in the eye, which can damage the optic nerve. Daily use of medicated eyedrops preserves sight by reducing the pressure.
The patients tried different formulations of the same medication in both micro- and regular drops, which were about twice as large, for a week at a time. The researchers tracked the patients’ eye pressure and side effects, such as burning, stinging, itching and dryness.
Matthews has health insurance which pays for his Azopt prescription drops, but those without insurance pay about $295 per bottle.
Matt Roth for ProPublica
hide caption
toggle caption
Matt Roth for ProPublica
Their results were conclusive: Microdrops worked as well as larger drops to lower eye pressure. They also reduced some of the uncomfortable side effects of larger drops. And all the patients preferred using them.
York and two of his Alcon colleagues published their results in 1992 in the American Journal of Ophthalmology. Robin, who consulted on the research, was the principal investigator.
“The microdrop delivery system worked,” York said recently. The drop “was manufacturable. It reduced stinging and the amount of drug needed to produce the same biologic effect. Excess drug draining out of the eye would be significantly reduced.”
Worries That Little Drops Would Lead To Lower Sales
But his innovative solution ground to a halt when it came to getting it on the market.
Back in the early 1990s, Jerry Cagle was the head of product development at Alcon. Cagle, who has a doctorate in microbiology, retired in 2008 after 32 years at the company. He said the microdrop project failed because it raised too many questions—all of them about profits.
First, would competitors in the cutthroat eye care business undermine the project? One time, Cagle recalled, Alcon wrapped one of its bottles in foil to reduce evaporation, extending the life of the product. A competing company started a rumor that Alcon’s drops needed the foil because they were toxic. Alcon had to remove the foil. “No good deed goes unpunished,” Cagle said.
Second, if Alcon reduced the drop size on this product, would they have to do the same on the company’s other eyedrops?
And maybe most crucially, how would the microdrops affect sales? Microdrops, Cagle recalled, had “the potential to increase the use-life of a bottle by a factor of two,” which could cut sales in half. But if they raised the price on the bottle to recover revenue, Cagle said, “then what’s a competitor going to say? ‘Look at Alcon’s product. It’s twice as expensive as ours.'”
Alcon would also have to get Food and Drug Administration approval. Company-funded studies would need to prove to the agency that the smaller drop was just as effective as a larger drop.
So the project was killed, Cagle recalled, doomed by the cost of bringing the microdrops to market, combined with the risks of whether they would sell. “I’m a believer in small drops, don’t get me wrong,” Cagle said. “If this had been an innovation we thought would have increased Alcon’s sales, I think it would be in the marketplace today.”
Novartis, which now owns Alcon, didn’t want to talk about the microdrop study. When asked about the drop size, a spokesperson said the drops include a “margin of safety” to ensure patients get enough of the drug in their eyes.
Robin recalled a different response back in the 1990s when he urged Alcon executives to pursue the microdrop. It was, he said, like asking your wife if you could leave town for your anniversary or her birthday. “It was a dead issue,” Robin said. “They would say, ‘It’s not profitable. We’re going to sell less drugs.’ Very simple. Bottom line.”
Drugmakers Have Long Known Drops Were Oversize
Twenty-five years later things haven’t changed. Those in the eye care industry — doctors, pharmaceutical officials, researchers — know that eyedrops are much larger than the eye can hold.
But there’s little focus on the waste. Dr. Michael Repka, spokesman for the American Academy of Ophthalmology, said the drops have been larger than the eye’s capacity for the three decades he’s been in practice. While the focus has been on drop administration and ensuring patients can get refills, he said, the industry should be looking at drop size.
You might think the FDA would intercede, but the agency’s mission is the safety and efficacy of drugs, not prices or indirect costs due to waste.
Since Alcon’s mothballed research in the early 1990s, other studies have similarly found that most drops on the market are larger than necessary. A 2006 study published in the Journal of Ocular Pharmacology and Therapeutics, for example, said 15 microliter drops are as effective as large drops. “Smaller drops would be preferable to minimize systemic exposure and spilled or wasted medicine,” the study said.
Two of its authors worked for the pharmaceutical giant Allergan, which also funded the study. Allergan still doesn’t make any drops that are 15 microliters or smaller. The company declined to comment.
Another study published in May in the journal BMC Ophthalmology said “a significant portion of an eyedrop is wasted.”
Internal drug company documents and depositions unearthed in a recent court case in Illinois also suggest that companies have long known their eyedrops are bigger than human eyes can absorb.
A 2002 Bausch & Lomb memo said dropper tips “deliver drops which exceed that of which the physiology of the eye can retain.”
A 2011 Pfizer memo said: “The drop size is not a medical dosing issue because the human eye can only absorb 7 (microliters) of fluid.” Common drop sizes are between 25 and 56 microliters, the memo added.
And in a 2014 deposition, a scientist from Allergan acknowledged that the company studied glaucoma drops of 5, 10, 15, 20 and 30 microliters in size and found no statistically significant difference in the ability of the drops to reduce eye pressure.
None of the drug companies wanted to discuss these documents or why they haven’t pursued a smaller drop.
Even a drug industry consultant, Gary Novack, said it would be ideal to have a smaller drop with a higher concentration of medicine. But Novack, a pharmacology expert who helps companies shepherd products to market, doesn’t believe reducing the size of drops would lower health care costs. The drug companies, he said, would “acclimate,” raising prices by charging by dose instead of volume.
Big Drops Strain Pocketbooks
It isn’t hard to find patients who are feeling the cost of this waste.
David Zinke, 67, lamented on Facebook that he couldn’t afford the $185 per month drops his doctor had just prescribed.
He was shocked to hear about microdrops. He always thought “a drop was a drop.” Zinke said he was getting by on his Social Security plus what he made by selling fudge and driving for Uber in Tucson, Ariz., clearing about $1,500 a month. His budget doesn’t cover the brand-name drug, so every day he uses three less expensive bottles of drops.
Matthews’ prescription drops keep his eye pressure down. Not using the eyedrops could lead to blindness.
Matt Roth for ProPublica
hide caption
toggle caption
Matt Roth for ProPublica
“If we could deliver it in a smaller and more appropriate way,” he said, “then that little bottle of 2.5 milliliters would last me two months instead of one.”
In Baltimore, Matthews, the teacher who fears running out of his drops, carefully puts a single drop of Azopt into each eye twice a day to preserve his remaining eyesight. His glaucoma has left him almost completely blind in one eye, and partially blind in the other.
The drops allow him to continue to work as a teacher and watch his beloved Baltimore Orioles. Azopt is made by Alcon. Matthews has good insurance, so he doesn’t pay the $295 sticker price. But he can’t get a refill until after the insurer figures his supply should have lasted, so it’s critical not to waste a single drop.
But no matter how hard he tries, the drug gets wasted. Each drop is more like a milky “gob” that collects in the corner of his eye. “Sometimes I feel like I’m wiping half of it out,” he said.
As he talked about the size of eyedrops, Matthews grew more irritated. “This whole blind thing takes some getting used to,” he said. “If the maker of the medicine isn’t really looking out for me, that bothers me.”
ProPublica is a nonprofit newsroom based in New York. You can follow Marshall Allen on Twitter:@marshall_allen.
News Brief: Health Care Deal, Travel Ban Blocked By Hawaii Judge
Senators reached a deal to stabilize the Affordable Care Act insurance markets for 2 years. President Trump’s third executive order restricting travel from some countries to the U.S. has been blocked.
Senators Reach Deal To Stabilize ACA Insurance Markets For 2 Years

Sens. Patty Murray, D-Wash., and Chairman Lamar Alexander, R-Tenn., say they have a tentative agreement to appropriate the subsidies for the next two years, restore money used to encourage people to sign up for Affordable Care Act health plans, and make it easier for states to design their own alternative health care systems.
Tom Williams/CQ-Roll Cal via Getty Images
hide caption
toggle caption
Tom Williams/CQ-Roll Cal via Getty Images
Less than a week after President Trump said he is cutting off subsidies to health insurance companies, lawmakers say they have a deal to restore the money and take other actions that could stabilize insurance markets for next year.
Sens. Lamar Alexander, R-Tenn., and Patty Murray, D-Wash., say they have a tentative agreement to appropriate the subsidies for the next two years, restore money used to encourage people to sign up for Affordable Care Act health plans, and make it easier for states to design their own alternative health care systems.
Alexander said the idea is to stabilize the markets in the short term while Congress continues to debate long-term changes to the ACA.
“Over the next two years, I think Americans won’t have to worry about the possibility of being able to buy insurance in the counties where they live,” he said in a conversation with reporters at the Capitol on Tuesday.
He said he spoke with President Trump over the weekend and that Trump said he supports the idea. However, Senate Majority Leader Mitch McConnell, R-Ky., would not commit to bringing the bill up for a vote, as Murray and Alexander seek enough support to ensure it could pass. The bill would need 60 votes to get across the finish line and would require at least a dozen Republicans to take a difficult political step in voting to shore up parts of Obamacare.
Trump, during a news conference with Greek Prime Minister Alexis Tsipras Tuesday in the Rose Garden, declared the Affordable Care Act “virtually dead” but said he supports the Alexander-Murray deal.
“It’s a short-term solution,” Trump said in response to a question about the deal. “The solution will be for about a year or two years and it will get us over this intermediate hump.”
The deal comes just days after the president said he is cutting off reimbursements to insurance companies for discounts they are required by law to give to low-income customers to reduce the burden of copayments and deductibles. They’re known as “cost-sharing reduction” payments.
The deal reached Tuesday, if it becomes law, would restore those payments for at least the next two years.
The agreement, which Murray says has not yet been finalized, would also make it easier for states to get waivers so they can set up alternative health insurance systems and offer a wider variety of insurance policies.
Alexander said those plans would retain the minimum coverage requirements under the ACA but could vary the definition of what is affordable. The deal also allows more people to buy so-called catastrophic plans that cover major illness but pay for less routine care.
“This is a small step,” Alexander said. “I’d like to undersell it rather than oversell it.”
An analysis by Charles Gaba, who runs a website, ACASignups.net, that tracks enrollment and costs of ACA insurance, showed that premiums are set to rise an average of 30 percent next year, with premiums in some states, including Iowa, rising much more.
As premiums rise, consumers who don’t think they need much health care are less likely to buy coverage, leaving sicker people in the market. Because those sicker customers spend a lot of money on care, they drive premiums up even higher. With restoration of the cost-sharing subsidies, those premium increases could be far smaller.
For much of this year, Trump had been threatening to cut off the subsidies. So insurance companies in many states filed paperwork to sell policies on the ACA exchanges next year that included two sets of premiums — one if the cost-sharing payments continued, and another if they were cut off. The deadline to finalize those prices was in late September, however, and it’s not clear companies will be allowed to cut their prices if the Alexander-Murray deal becomes law.
The agreement would also restore $106 million in money to publicize the open enrollment period for ACA health insurance. Trump had cut the budget for ACA outreach by 90 percent earlier this year, a move many advocates said would suppress enrollment.
Lori Lodes, who ran ACA outreach during the Obama administration, said that money will only be effective if it is approved early enough to reach those who need coverage. Open enrollment on the federal exchange begins on Nov. 1 and ends on Dec. 15.
Murray said she and Alexander had had discussions with more than half the Senate about the bill and she believed the plan would get broad support. Alexander said they’ll work to get co-sponsors for the legislation throughout the week so they can bring a bill to McConnell for consideration. McConnell has not said whether he supports the effort.
After Taking On Big Tobacco 20 Years Ago, Former Mississippi AG Is Trying Again With Opioids
When Mike Moore was Mississippi’s attorney general, he spearheaded the 50-state lawsuit against Big Tobacco. Now, he’s trying to do the same thing against opioid manufacturers and distributors.
Trump Still Hasn't Fulfilled Promise To Declare Opioid Epidemic As A National Emergency
President Trump’s nominee to head the Drug Enforcement Administration pulled out amid controversy. This leaves Trump without a permanent heads at Department of Health and Human Services and the DEA. All the while, his promised declaration of a national emergency on opioids has yet to materialize.
If You Want An IUD, Take Note Of Trump's New Birth Control Policy

Most employers are likely to continue paying for birth control for women. But there are exceptions.
Science Photo Library/Getty Images/Science Photo Libra
hide caption
toggle caption
Science Photo Library/Getty Images/Science Photo Libra
The Trump administration has made a number of changes to health policy in the past two weeks, raising questions about how consumers will be affected. Will the new rules for birth control coverage affect access to an IUD? Might an association health plan help bring down costs for workers at small businesses? And if you’re healthy, doesn’t a short-term health plan that is cheaper than marketplace coverage make sense? Here are some answers to those questions.
IUDs are expensive. Will they be unaffordable under the new contraceptive rules announced by the Trump administration? Should people make an appointment to get one now before employers change their minds about coverage?
There’s probably no need to rush out to your doctor’s office to get an IUD, but you should keep an eye on this issue.
Under the Affordable Care Act, most health plans are required to cover all methods of birth control approved by the Food and Drug Administration without charging women anything for them. Religious employers and some private employers with strong religious objections are exempt from the requirement, but it’s a pretty limited group. Or it was until the Trump administration issued new rules during the first week of October that open the door for many companies or nonprofit organizations with religious or moral objections to contraception to stop offering it.
“These new exemptions are sweeping,” says Adam Sonfield, a senior policy manager at the Guttmacher Institute, a reproductive health research organization. Essentially any employer will be able to claim an exemption from the birth control coverage requirement, he says, and there are no provisions to appeal it.
What’s not at all clear, however, is how many employers will take advantage of the new rules. The administration says it expected the number to be small, probably just the companies that had brought suit against the old rules.
Even before the ACA passed, many plans covered contraception. They didn’t necessarily cover all FDA-approved methods, however, and women typically had to pay a share of the cost.
Several states have laws that protect birth control coverage, says Mara Gandal-Powers, senior counsel at the National Women’s Law Center. Some require that if a plan offers prescription drug coverage, for example, it must cover contraceptives. Others have adopted the ACA rules that require coverage of all FDA-approved methods without cost sharing. But those laws would not apply to employers that pay their employees’ health care claims directly rather than buy state-regulated insurance.
The protection from cost sharing is key, experts say, especially for highly effective methods like the IUD, which might cost $1,000 upfront.
“Even if you’ve met your deductible, if you have 20 percent coinsurance that’s $200, and for many people that’s not feasible,” Gandal-Powers says.
If your employer does decide to stop providing insurance coverage for contraception, in most cases plans have to give workers 60 days’ advance notice of the benefit change, giving you time to get that IUD if you decide to.
Association health plans, which the Trump administration is encouraging, seem like kind of a good idea. If more small companies band together, won’t that make coverage cheaper?
President Donald Trump signed an executive order last Thursday that directs several federal agencies to consider proposing rules that, among other things, would allow more employers to buy health insurance through associations.
If you’re young and healthy, getting coverage through an association health plan might indeed be cheaper. But you’ll likely forgo consumer protections that are required for plans sold on the individual and small-group markets, says Kevin Lucia, a research professor at Georgetown University’s Center on Health Insurance Reforms.
Plans currently sold in those markets have to cover 10 so-called essential health benefits. Association health plans would likely sidestep that requirement. (However, implementing federal rules for these plans will take the administration some time, so it’s not clear if or when they might be expanded.)
“If you don’t cover maternity, mental health or hospitalization, the premiums are going to be lower,” Lucia says.
The administration’s press release about the executive order says that employers couldn’t exclude any employees from joining association health plans or “develop premiums based on health conditions.”
But association health plans have been known to use many strategies to cherry-pick employers with healthy workforces, Lucia says.
“The risk is that each individual small-employer’s rates would be separately determined based on its employees’ medical claims, potentially splitting the market into employers with sicker workers and those whose workers are healthier,” he says.
Short-term plans are cheaper than Obamacare plans. If people don’t have pre-existing conditions and are willing to pay the penalty under the law for not having minimum coverage, what’s the downside?
Trump’s executive order that encouraged the expansion of association health plans, discussed above, also aims to expand the availability of short-term plans. Under Obama administration rules, the coverage period of short-term plans was limited to less than three months. This executive order proposes to expand that, perhaps to just under a year.
In addition to not covering preexisting conditions, short-term plans often exclude certain types of coverage, such as prescription drugs and maternity, and impose dollar limits on coverage. The maximum out-of-pocket spending limits are often higher than coverage in a marketplace plan too.
The potential downside is the possibility that you may develop a medical condition or have an accident that requires expensive medical care while you’re covered under this plan, experts say.
“If the reader has a car accident, the insurer wouldn’t renew the policy,” says Timothy Jost, a professor emeritus of law at Washington and Lee University in Virginia who is an expert on health law. “If coverage is terminated, you’re not eligible for a special enrollment period [on the exchange]. So you could just get marooned.”
Kaiser Health News is an editorially independent news service that is part of the nonpartisan Henry J. Kaiser Family Foundation. Follow Michelle Andrews on Twitter: @mandrews110
Washington Post, 60 Minutes Investigation Finds Bill Helped Fuel Opioid Crisis
NPR’s Kelly McEvers talks to Scott Higham of The Washington Post about the paper’s investigation of drug industry efforts to lobby the Drug Enforcement Administration and Congress to weaken enforcement on opioid abuse.
Iowa Tries To Prevent Health Insurance Premiums From Escalating
The White House has canceled a subsidy to insurance companies. Iowa’s Health Insurance Commissioner Doug Ommen discusses the state’s plan that seeks to lower costs for tens of thousands of residents.