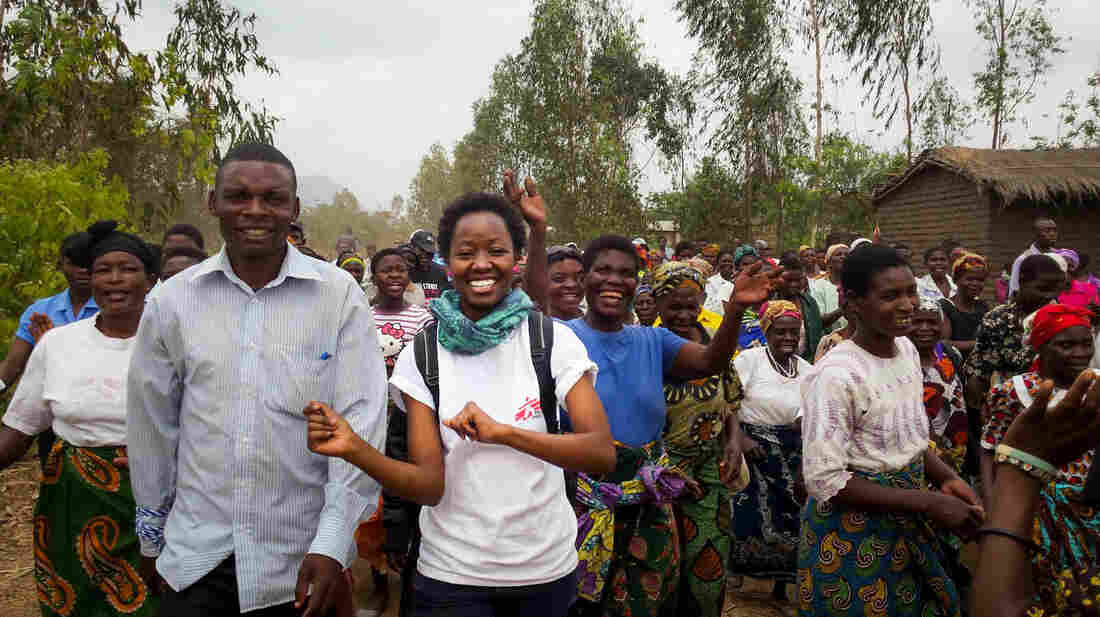An STD testing kit from myLAB Box allows users to gather samples at home and mail them back to the company.
Courtesy of myLAB Box
hide caption
toggle caption
Courtesy of myLAB Box
America is losing the battle against sexually transmitted infections. Cases of chlamydia, gonorrhea and syphilis all hit record-high numbers in 2015. Tens of thousands contract HIV every year in the U.S., and oral cancers caused by human papillomavirus are increasing.
So startups are popping up online to help serve what they see as unmet demand for STD testing. One advertises that you can “get a sexy deal” by ordering.
The question is whether those companies can survive — at least one left the market before its product even launched — and whether the services they offer get the right tests to the right people.
Although encouraging people to get tested is a simple enough public health message, that doesn’t mean it’s simple to carry out, says Kevin Ault, a professor of obstetrics and gynecology at the University of Kansas Medical Center in Kansas City, Kan.
“You have to make the appointment at the doctor’s office, drive to the doctor’s office, give the sample to the doctor, the doctor sends it to the lab, you wait for the results to come back, and then you wait for the doctor to call you,” Ault says. And the Centers for Disease Control and Prevention recommend that people in some at-risk groups do all that several times a year.
“The biggest advantage of home tests in general is if you catch HIV or chlamydia early on, you can change the natural course of the disease,” Ault says.
Few options exist to make the process easier. So far, there is just one test approved that gives rapid results in the home, and it’s for HIV. The startups are hoping that being able to collect samples at home will be enough to encourage people to get tested.
“The concept of providing the possibility of a self-sampling approach to test for STDs is really going to be our future in terms of diagnostic testing,” says Jennifer Smith, an associate professor of epidemiology at the University of North Carolina Gillings School of Global Public Health in Chapel Hill. Hill also consults for myLAB Box, one of companies offering these services. “Getting the actual test to the patient is going to be a way of not only increasing access and improving acceptability, but also cutting down on unnecessary medical visits,” Smith says.
Big increases in people infected with STDs
A sobering report from the CDC last October revealed just how much new approaches are needed to combat increasing infection rates. More than 1.5 million people contracted chlamydia in 2015, an increase of 5.9 percent from the year before. Similarly, gonorrhea cases jumped 12.8 percent to almost 400,000 cases. The nearly 24,000 new cases of primary and secondary syphilis (the two most infectious disease stages) represented a 19 percent increase.
Aside from early symptoms that several STDs can cause — such as painful urination, discharge, bleeding, swelling or pain — long-term symptoms in people who don’t receive treatment can be serious. Untreated gonorrhea, for example, can cause infertility and long-term pelvic or abdominal pain in men and women as well as ectopic pregnancies, which can be fatal. If syphilis is not treated, it can damage the brain, eyes and nervous system, potentially resulting in severe headaches, poor muscle coordination, paralysis, numbness, dementia or blindness. In rare cases, syphilis can cause death 10 to 30 years after infection.
STDs also have downstream consequences for the next generation. Cases of congenital syphilis, which can cause severe illness and stillbirth, has increased in newborns 38 percent from 2012 to 2014, according to the CDC, even though women don’t even represent 10 percent of new infections overall.
New HIV infections have been steadily dropping, but 2015 still saw more than 39,500 new cases. And although HPV, the most commonly transmitted STD, resolves on its own in most people, it still causes about 31,500 new cancers annually.
“When something affects millions of people, even a low rate of serious outcomes translates to a lot of people,” says H. Hunter Handsfield, a professor emeritus at the University of Washington who consults for the CDC on STDs and spent a quarter of a century directing the STD control program for Seattle’s public health department. “We have an ongoing and important public health problem of people getting HIV and getting cervical and other HPV-related cancers. The numbers of those actual cancers are small, but that’s a big deal for each of those people.”
Startups See An Opportunity
The idea of online STD testing isn’t new, but most services so far have been localized, limited in test options or still require visiting a lab or pharmacy.
For example, residents of Maryland, Washington, D.C., and Alaska can request kits to be mailed to them with self-collection instructions and materials for genital and/or rectal swabs, but only for gonorrhea, chlamydia and trichomoniasis. Planned Parenthood has begun offering similar services but only for gonorrhea and chlamydia and only in Idaho, Minnesota and Washington. More than a dozen commercial companies nationally let consumers order STD testing kits online for other infections, but buyers still have to visit a local lab for sample collection.
The online businesses aim to offer many more tests without customers needing to go anywhere except the mailbox. The two business models are subscription-based or one-off orders: Consumers order the test, receive it in the mail, collect their own blood, urine, genital and/or rectal samples, mail samples back in a prepaid envelope and then wait until results are available to check online.
One company, GetTested, still has a live website but has ceased operation, according to a spokesperson. Another, Mately, doesn’t appear operational and did not respond to multiple attempts to request an interview. That leaves myLAB box, which has been tweaking its services and procedures since its launch in December 2013 as executives learn what does and doesn’t work.
The CDC generally supports the idea of at-home STD testing, according to John Papp, a microbiologist in the CDC’s Division of STD Prevention and author of CDC’s lab testing recommendations for gonorrhea and chlamydia.
“From our perspective in public health, we want people to have access,” Papp says. “The concept of greater access, however that looks, if it’s by a website or a van down by the river, is always a good thing. But the regulatory piece needs to be adhered to.”
But little regulation exists for online, at-home STD testing. The labs where tests are performed should meet the standards of the Clinical Laboratory Improvement Amendments, and the tests themselves should be FDA-approved when available. The tests offered by myLAB Box meet both those requirements.
“There’s no FDA indication for at-home collection and sending it into a laboratory,” Papp says. “Having said that, if the specimen is collected properly, regardless of the setting, the test is probably being performed adequately.”
Most of the tests detect some piece of the organism itself. Three others, for hepatitis C, syphilis and herpes simplex type II, test for the body’s antibodies made in response to the infection. The HIV test looks for both the virus and antibodies. The tests have been shown to work even with samples exposed to extreme temperatures, so having a blood spot and urine sample sitting in a Florida mailbox in August shouldn’t affect results, Handsfield says.
Reaching the right people
But Handsfield says online tests don’t reach the people who need testing and treatment the most.
“It’s a good idea, with a giant caveat that it reaches the wrong people,” Handsfield says about online services. “The highest infection rates are in people with lesser education or lower income, in inner cities or the rural kid in a red state immersed in a methamphetamine world.”
He would like to see public health departments partner with online sites and subsidize the cost to promote home self-testing for a broader population of high-risk, lower-income people. The CDC’s October report, for example, showed that 15- to 24-year-olds make up half of gonorrhea cases and almost two-thirds of all chlamydia ones. “These are not the same people who are paying money to buy tests online,” Handsfield says.
MyLAB Box offers three pricing tiers: the “Safe Box” for $189 (HIV, chlamydia, gonorrhea and trichomoniasis); the “Uber Box” for $269 (adds hepatitis C, herpes simplex type II and syphilis); and the “Total Box” for $399 (adds HPV, Mycoplasma genitalium and ureaplasma plus rectal and throat testing for chlamydia and gonorrhea). Each test can also be purchased individually for $79.
But Gary Richwald, myLAB Box’s medical director and chief scientific officer, says the company is reaching the right people. He says their rates of positive tests are on par with or higher than what he saw when he ran STD clinics for Los Angeles County, the largest such program in the U.S., from 1989 to 2000. For example, 7.3 percent of myLAB Box clients’ tests for chlamydia were positive in February. Community rates at L.A. clinics two decades ago, where the population would presumably have been high risk, ranged from 4 percent to 5 percent, Richwald says, with family planning clinic rates lagging just behind that.
“The data show in every study that people who voluntarily go somewhere to be tested have higher rates than the general population who might be tested door to door,” Richwald says. And yet “the vast majority of people with STDs never get tested, and they are the principal source of new infections.”
Richwald describes the company’s customer base as people mostly in their mid- to late 20s, with many in their 30s and 40s as well, and often at a transitional stage in their life, such as having recently ended a relationship or gotten divorced. Economically, they seem to hover between lower middle class and middle upper class, he says. Customers include residents of areas with doctor shortages, where getting tested requires going to urgent care or the ER; single mothers without time to get to a clinic or doctor’s office; and individuals with previous unsatisfactory health care experiences.
They also tend to have three other characteristics: comfort and familiarity with using the Internet, a desire for convenience — “I can’t tell you how many people said they collected their specimen after midnight,” Richwald says — and concerns about privacy.
“With this election and general concerns about privacy in this country, people are afraid that even their request for a test, much less their positive, would end up in some place that collects health-related information,” Richwald says. A number of customers include those in the health care field themselves, he says, such as physicians, dentists and nurse practitioners.
Choosing the right tests
One big question is what to test for.
Public health clinics generally offer free testing of gonorrhea, chlamydia, syphilis and HIV, with some offering various additional tests, such as trichomoniasis, HPV or herpes type II. Few public health clinics test for ureaplasma, hepatitis C or Mycoplasma genitalium.
A person’s first instinct may be to test for “everything,” especially if they have a sexual history or recent sexual experience that could be a concern. But not everybody should be tested for every infection.
Hepatitis C, for example, is currently among the tests offered by myLAB Box, but it’s not considered a sexually transmitted disease for anyone other than men with HIV who have sex with men, Handsfield says. (All individuals born between 1945 and ’65 are recommended to be tested once for hepatitis C, however.) And men are not typically tested for HPV because no treatment exists for the infection, and it’s unclear what to do with a positive result.
MyLAB Box company co-founder Lora Ivanova says the company trusts the consumer to do the homework on what tests to order.
“Our role is to make it as easy as possible for the person who has decided to get tested to get the test they want,” Ivanova says. “For a long time, consumers have been limited to the tests they can take based on the medial debate. We’re taking the position that the consumers ultimately have the right to know. We don’t see why we as providers should limit their access to care.”
But if doctors and public health policymakers cannot agree on who should be tested for infections like Mycoplasma genitalium and ureaplasma, Handsfield says, then how would a consumer make that decision?
“The issue of who to test and what tests to do continues to be a question that’s very important but does not have an exact answer,” Richwald acknowledges. That’s partly why he was brought on, and Ivanova did say the company’s system “is in constant flux” based on “recommendations and available data.” The company doesn’t offer testing for herpes simplex type I, for example, because 60 percent to 70 percent of individuals already have antibodies, acquired non-sexually in childhood.
Richwald also says the company isn’t testing for ureaplasma anymore, but the test still appears on the company website. Mycoplasma genitalium presents a conundrum as well.
First, no FDA-approved diagnostic test for the bacteria exists. Experts disagree on how to interpret positive results, Handsfield says. It’s a common bacteria found in about 1 percent of the population, but most people don’t have symptoms. Treatment is challenging and not recommended for infections without symptoms. But for those with symptoms — vaginal pain or itching, discharge from the urethra, painful urination and painful or swelling joints — treatment can prevent pelvic inflammatory disease or worsening symptoms, Richwald says.
Another consideration people must weigh is when to test, because incubation periods vary by disease. If someone has been regularly sexually active, especially without using a condom, and has not been tested in the six months, timing is less relevant. But if someone is testing after a specific encounter, some infections, such as HIV, cannot be detected immediately.
“Often people get tested too soon, such as a week after exposure,” Handsfield says. Chlamydia and gonorrhea can usually be detected after several days (a week on the conservative side), but herpes and one HIV test require up to three months of delay before testing.
A chart on the myLAB Box site provides time frames for testing and, when necessary, retesting. It recommends that people wait until the end of the time frames listed before testing unless the person plans to retest. The ideal testing window for Mycoplasma genitalium, however, is unknown, Handsfield says.
“For the panel as a whole, I would say wait three months if you have no symptoms,” Handsfield says. “If you have symptoms — if you’re having urethral discharge, unexplained vaginal discharge, abdominal pain — online testing is not for you. You need to see a doctor.”
Handling positive tests, whether true or false positives, also requires careful consideration. The newest syphilis tests, for example, are known for giving a lot of false positives, Handsfield says, and that can lead to increased anxiety between a first test and a retest, although the same concern would exist at a community clinic. At myLAB Box, Richwald personally calls all customers with a positive HIV result and ensures they get an appointment with an HIV specialist group. Immediate treatment can dramatically reduce their infectiousness while improving their health, he says. A positive result for syphilis requires confirmation at a clinic in person, and someone with chlamydia and symptoms of pelvic pain, for example, would be told to go to a clinic or urgent care.
MyLAB Box regularly reviews new research to inform their decisions, but it remains a tricky line to walk: making tests widely available to the public while trying to guide them toward the best tests for their situation without driving them away.
“There’s a lot of fear, hesitation and confusion, and I think what it has ultimately done is turn people to the point where they’re sweeping it under the rug,” Ivanova says. “At the end of the day, it’s about getting the person to get tested. If we lose that one single time in a year or in two or five years that they have mustered the courage to get online and get the tests, they might spend the next five years infecting every partner they have.”
Tara Haelle is the co-author of The Informed Parent: A Science-Based Resource for Your Child’s First Four Years. She’s on Twitter: @tarahaelle
Let’s block ads! (Why?)